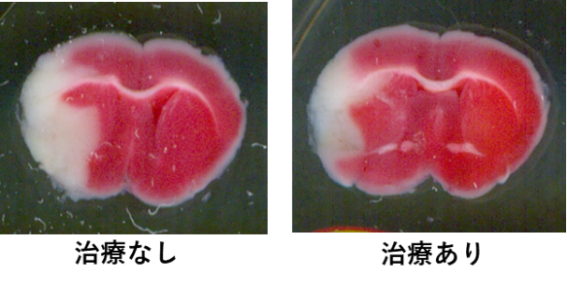
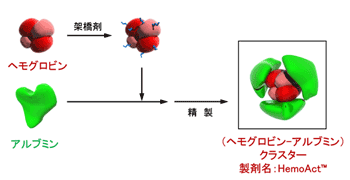
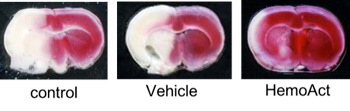
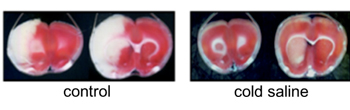
Sphingolipid as a neuroprotective agent for ischemic reperfusion injury
Sphingolipids have long been viewed as merely ubiquitous components of the cell membrane. Recently, sphingolipids have been increasingly revaluated because they are now recognized to not only regulate vital cell functions, but also form cell membrane microdomain “lipid rafts” for integrating cell signalling. FTY720 (Fingolimod) is a novel immunomodulatory agent, which in its phosphorylated form acts as a high affinity agonist of the Sphyngosine-1-Phosphate (S1P). It became the first oral drug to be FDA-approved for clinical use in the treatment of multiple sclerosis. FTY720 readily crosses the blood-brain barrier and exerts a number of direct effects in the central nervous system. FTY 720 is phosphorylated by SphK, which then acts on 4 of the 5 known S1P receptor subtypes (S1P1, S1P3, S1P4, S1P5), and shows neuroprotective effect against many central nervous system disease including cerebral ischemia. We are focusing on the microglical effect of the FTY720 and try to expand the clinical use of this drug for ischemic reperfusion injury.